How to Balance NaHCO3 + HCl = NaCl + CO2 + H2O (Sodium Bicarbonate Plus Hydrochloric Acid) - YouTube

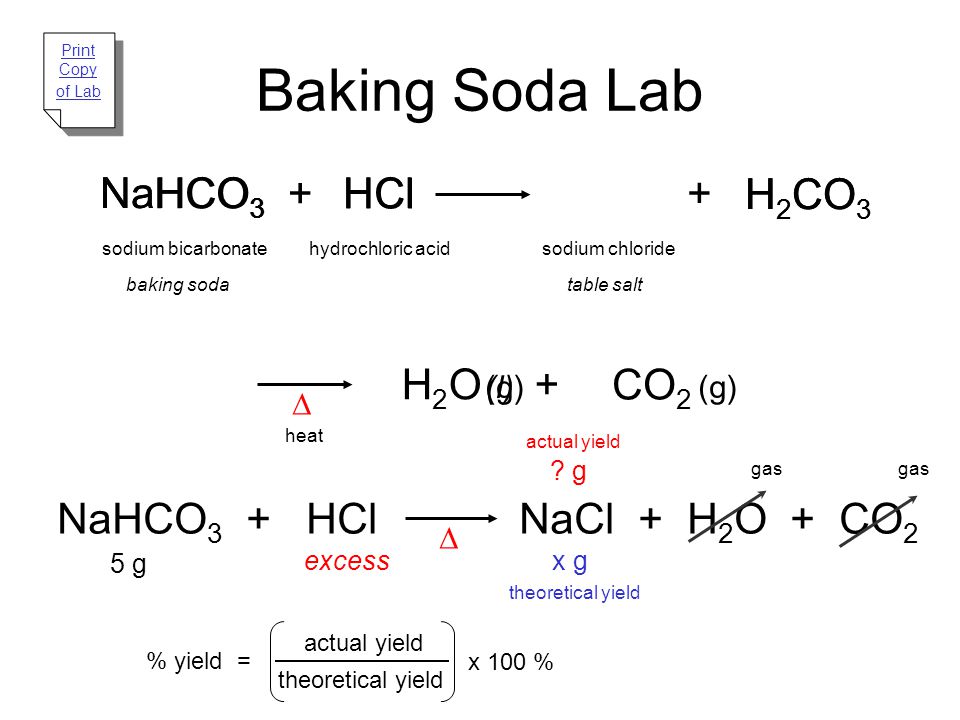
HCl + Baking Soda Stoichiometry Expt Sp2022 - Stoichiometry I: Theoretical, Actual and Percent Yield - Studocu

Hydrochloric acid reacts with baking soda. A 0.1M solution of hydrochloric acid (HCl) is added to a beaker with solid baking soda (sodium bicarbonate, NaHCO3). Carbon dioxide bubbles are produced: NaHCO3 +

MOST COST-EFFECTIVE BRAND OF BAKING SODA Keerthana Sundar, Deepa Selveraj, Desiree Chew and Amanda Lee from ppt download

SOLVED: Baking soda or sodium hydrogen carbonate (NaHCOs) solid is reacted with concentrated hydrochloric acid at 37 %Cand 1.00atm according to the equation: NaHCOs (s) + HCI (aq) NaCl (aq) - COz (
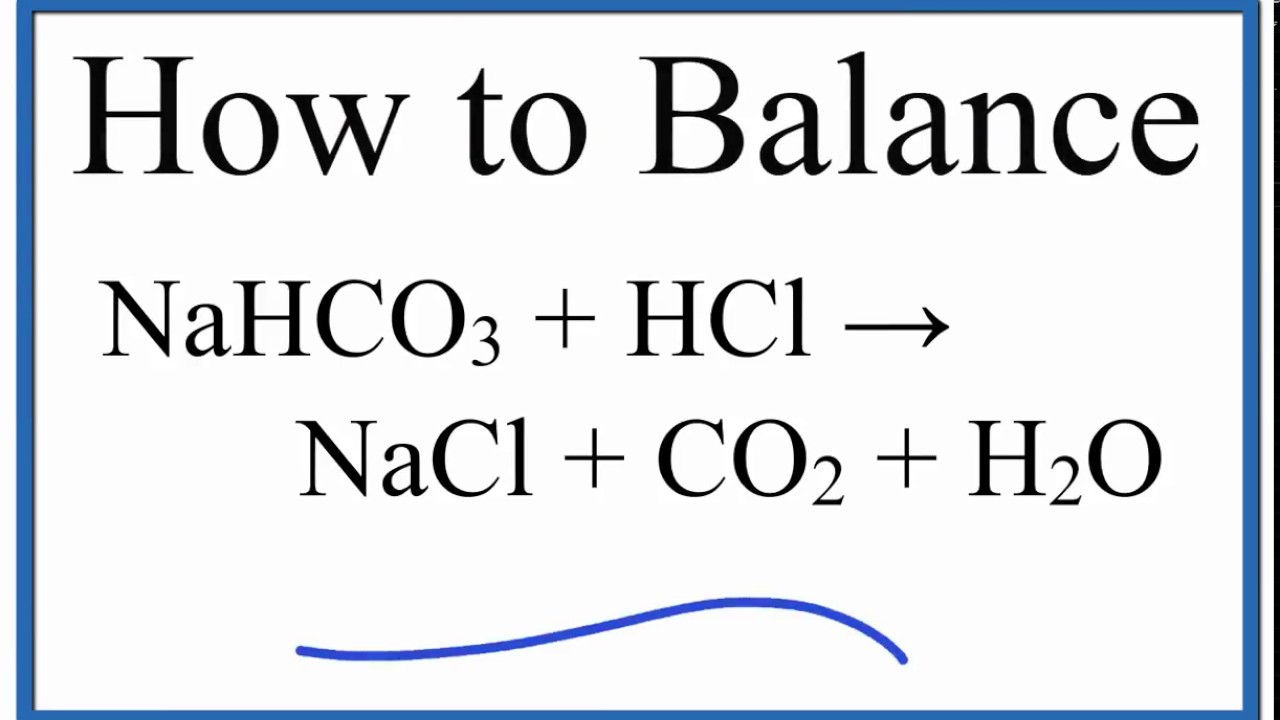
How to Balance NaHCO3 + HCl = NaCl + CO2 + H2O (sodium bicarbonate plus hydrochloric acid) - YouTube

Hydrochloric AcidBaking SodaSodium ChlorideCarbon Dioxide Theoretical Predictions Molar Mass Hydrochloric Acid Baking Soda Sodium Chloride Carbon Dioxide. - ppt download