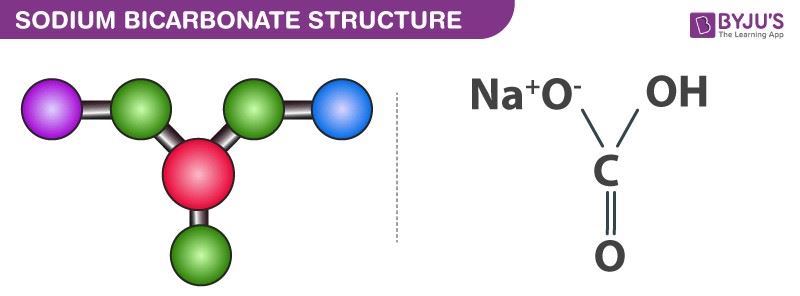
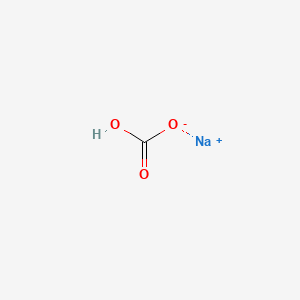
formula unit mass and molecular formula of baking soda and gypsum - Science - Atoms and Molecules - 8791425 | Meritnation.com

SOLVED: It is possible to make sodium carbonate (molar mass: 105.99 g/mol) from baking soda (sodium bicarbonate molar mass: 84.006 g/mol) by the following reaction: 2 NaHCO3(s) + NazCO3(s) COz(g) HzO(g) You

Hydrochloric AcidBaking SodaSodium ChlorideCarbon Dioxide Theoretical Predictions Molar Mass Hydrochloric Acid Baking Soda Sodium Chloride Carbon Dioxide. - ppt download

1. Calculate the molecular mass of the following: (a) Baking soda (b) Washing soda (c) Gypsum (d) Quick - Brainly.in

Write the formulae for the following and calculate the molecular mass of each one of them.(a) Caustic potash(b) Baking powder(c) Limestone(d) Caustic soda(e) Ethanol(f) Common salt

Write the formulae for the following and calculate the molecular mass for each one of them - YouTube

SOLVED: Baking soda and vinegar react according to the following chemical equation: NaHCO3+C2H4O2→CO2+H2O+NaC2H3O2 Determine how much vinegar would be needed to react with 9 grams (approximately 1 tbsp.) of baking soda by
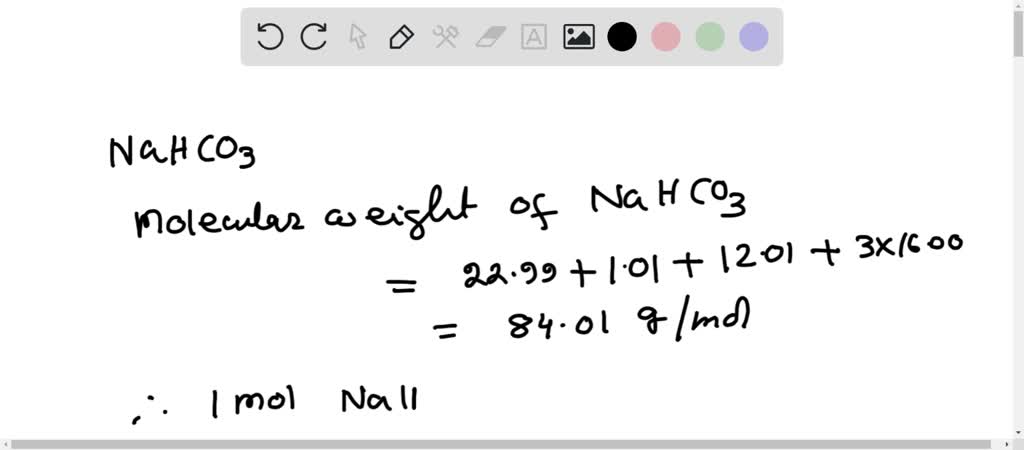
SOLVED:Calculate the number of grams of baking soda, NaHCO3, that contain 12.2 moles of carbon. Baking soda