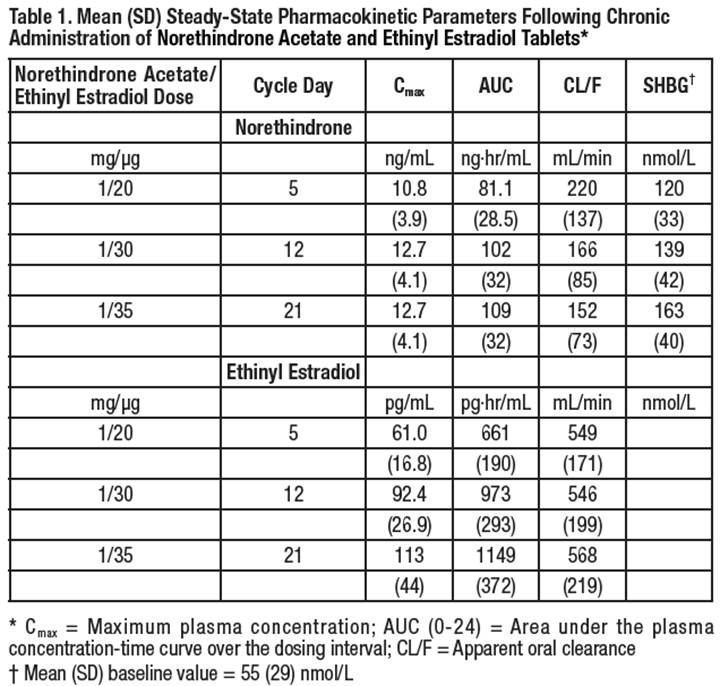
Tilia® Fe(Norethindrone Acetate and Ethinyl Estradiol Tablets, USP and Ferrous Fumarate Tablets*)1 mg/0.02 mg, 1 mg/0.03 mg, 1 mg/0.035 mg*Ferrous fumarate tablets are not USP for dissolution.

Esterified Estrogens and Methyltestosterone Tablets, 1.25 mg/2.5 mg Esterified Estrogens and Methyltestosterone Tablets, 0.625 mg/1.25 mg (Half-Strength) CIII