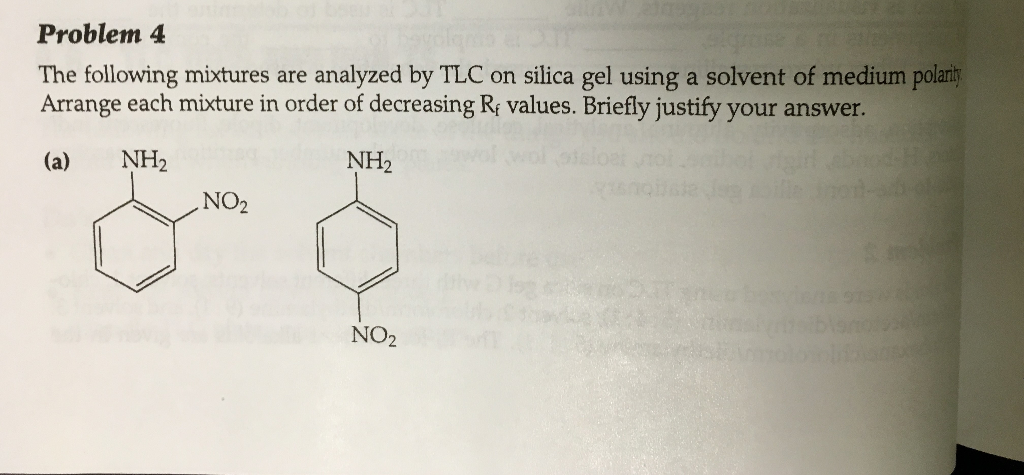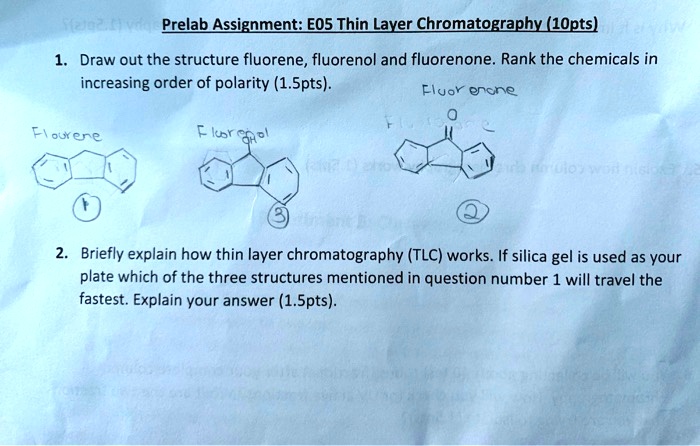
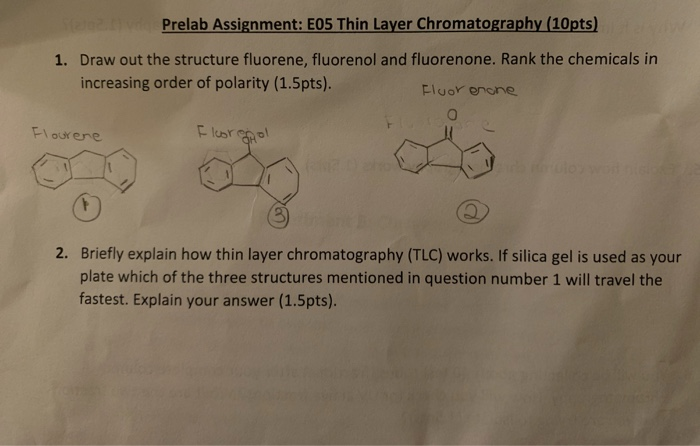
SOLVED: Prelab Assignment: EOS Thin Layer Chromatography (1Opts) Draw out the structure fluorene, fluorenol and fluorenone: Rank the chemicals in increasing order of polarity (1.Spts). Fluor encne Icbt 9 Briefly explain how

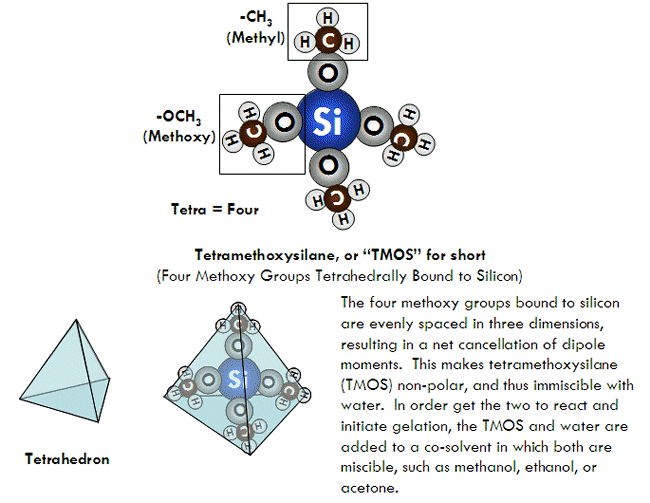
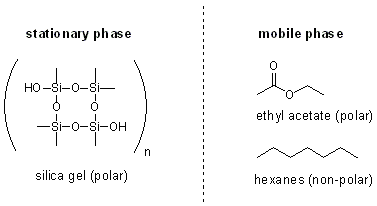
SOLVED: Define chromatography: Given the structure of silica gel below, calculate the Si-0 bond polarity using electronegativity values (Zumdahl, Sec 8.2). Is silica gel polar or nonpolar? Silka ctlcnded utnulun and autlect
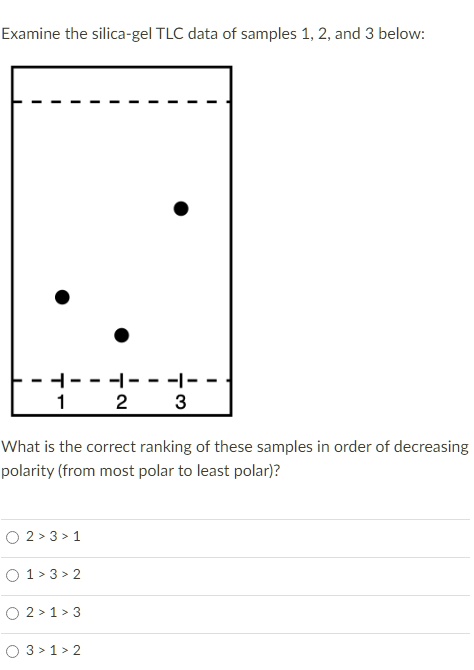
SOLVED: Examine the silica-gel TLC data of samples 1,2, and 3 below 2 3 What is the correct ranking of these samples in order of decreasing oolarity(from most polar to least polar)

Consider the following silica gel TLC plate of compounds A, B, and C developed in hexane and answer the following questions. a) Determine the Rf values of compounds A, B, and C (

Thin-Layer Chromatography (TLC) Uses: To separate the components of a mixture To determine the purity of a compound To see if two compounds are identical. - ppt download

Preparation of mixed-mode stationary phase for separation of peptides and proteins in high performance liquid chromatography | Scientific Reports

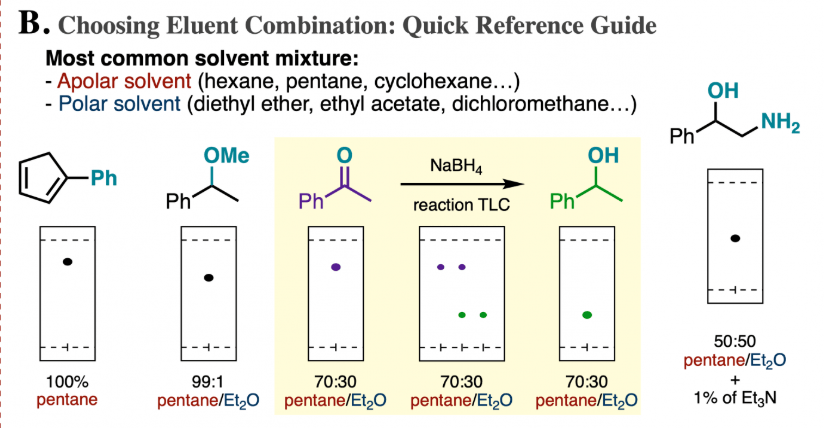
organic chemistry - How does the polarity of the eluent and sample affect the Rf value in thin layer chromatography? - Chemistry Stack Exchange

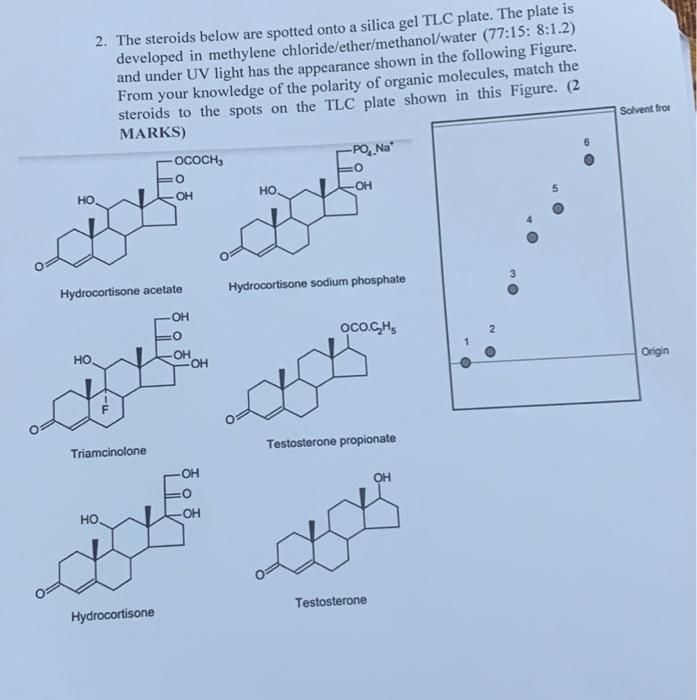



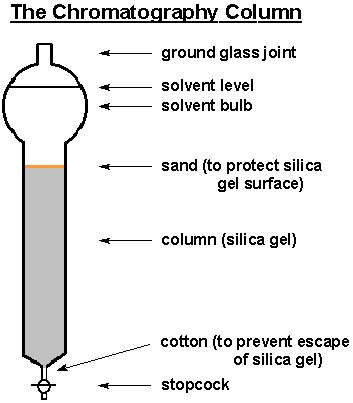
.png?revision=1&size=bestfit&width=1103&height=424)