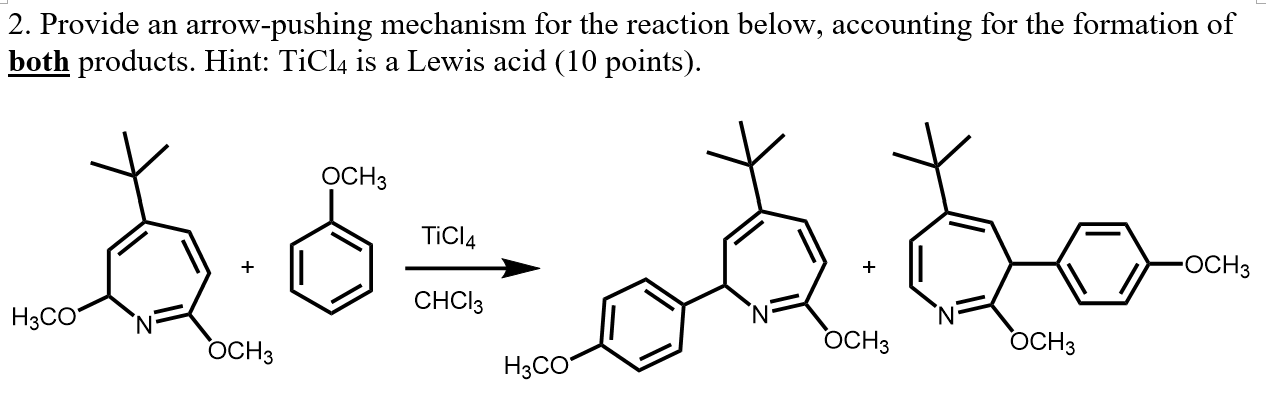
TiCl4-Promoted Baylis−Hillman Reaction: Mechanistic Rationale toward Product Distribution and Stereoselectivity | The Journal of Organic Chemistry

Table 2 from Accurate experimental and theoretical enthalpies of association of TiCl4 with typical Lewis bases used in heterogeneous Ziegler-Natta catalysis. | Semantic Scholar

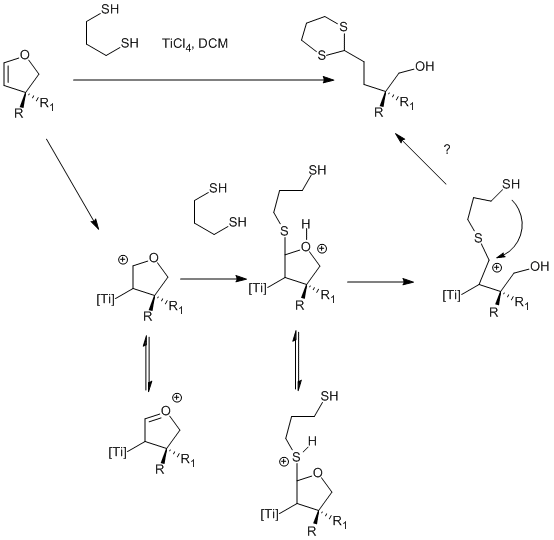
TiCl4‐Et3N‐mediated one‐step synthesis of γ‐alkylidenebutenolides from ketones: Application to natural product synthesis - Li - 2020 - Journal of Heterocyclic Chemistry - Wiley Online Library
![TiCl4: An efficient catalyst for one-pot synthesis of 1,2-dihydro-1-aryl-naphtho-[1,2-e][1,3]oxazin-3-one derivatives and their drug score analysis - ScienceDirect TiCl4: An efficient catalyst for one-pot synthesis of 1,2-dihydro-1-aryl-naphtho-[1,2-e][1,3]oxazin-3-one derivatives and their drug score analysis - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S1878535213001949-gr2.jpg)
TiCl4: An efficient catalyst for one-pot synthesis of 1,2-dihydro-1-aryl-naphtho-[1,2-e][1,3]oxazin-3-one derivatives and their drug score analysis - ScienceDirect

TiCl4‐Mediated Dealkylative Cyclization of Arylidene‐azol‐ones - Zaitseva - 2022 - ChemistrySelect - Wiley Online Library



.jpg)